There appears to be some consensus to wait 30 days before adding salt to new plaster pools. Yet some in our industry claim that it is okay to add salt within a couple of days of filling the pool. So, who is right?
To answer that question, the onBalance team conducted an experiment in a laboratory setting to determine what effects salt may have on fresh plaster and on the curing process. We compared adding 3000 ppm of salt at the beginning of the start-up process with not adding salt.
The results obtained suggest that adding 3000 ppm of salt does have a negative effect on plaster when added at startup, and up to two weeks.
We observed that when salt is added to balanced water containing fresh plaster coupons, the pH of the water rises very quickly and much higher than normal. Even after adding acid repeatedly to lower the pH, the pH rises again quickly and generally above 8.5. This indicates that calcium hydroxide (a plaster component) is being dissolved and removed (uniformly) from a plaster surface when salt water concentration is present.
That effect causes porosity of the plaster finish, which weakens and ages the surface. Adding salt at start-up also makes it more difficult to get the pH down and balance the pool water during the start-up period.
It was also observed that the negative effect (increased calcium hydroxide dissolution and pH rise) of adding salt to new plaster pools only lasts about two to three weeks. That is because a very thin layer of the new plaster coat (immediate surface) typically becomes “carbonated” (calcium hydroxide being converted into calcium carbonate) over the first two to three weeks after filling with balanced water.
This (carbonation) conversion creates a protective thin layer and more durable plaster surface. It appears that once a plaster surface has been sufficiently and properly carbonated, salt water (less than 5,000 ppm) no longer has the same negative effect.
Our experiment used well-made plaster coupons, which received proper curing, and positive Saturation Index water.
The bottom line. Waiting 30 days before adding any salt is recommended for plaster pools, including for quartz and pebble pools. And of course, when adding salt to the pool, never allow undissolved salt to lie and remain on the plaster surface for any length of time.
Additional information to help understand this issue.
Let’s consider what happens in the initial days and weeks of a new plaster surface. When plaster is applied and a new pool is filled, the cement portion of the new, hardened pool plaster contains about 10 to 15% calcium hydroxide, the rest being primarily calcium silicates, aluminates and carbonates. These latter products are durable and relatively insoluble in water.
However, calcium hydroxide, which has a pH of about 12, is softer, somewhat soluble, and can be dissolved from a plaster surface even by typical balanced tap water. Indeed, the Saturation Index as a maintenance tool to protect calcium carbonate is not applicable to fresh new plaster (i.e., less than a few weeks old) because the new plaster has some calcium hydroxide on the surface.
That is why the pH of the water in a freshly filled pool usually rises as the pool is being filled. High pH calcium hydroxide dissolves from the plaster surface and into the water, causing the pH to rise. But once the new plaster surface is carbonated (which means the calcium hydroxide is converted to calcium carbonate) that process stops.
Dissolved calcium hydroxide becomes converted to calcium carbonate in the pool water, and this is the source of so-called “plaster dust” in new plaster pools. When undergoing a traditional start-up process, carbonation of the surface usually lasts about 1½ to 2 weeks… which is why new plaster dust is generated for that long – and then stops. At that time the pH becomes stable in the pool, since new hydroxide is no longer being dissolved into the pool water.
If, in the field, proper plastering practices are not followed closely, which results in a lower quality finish, more time may be needed before salt should be added.
Experiment:
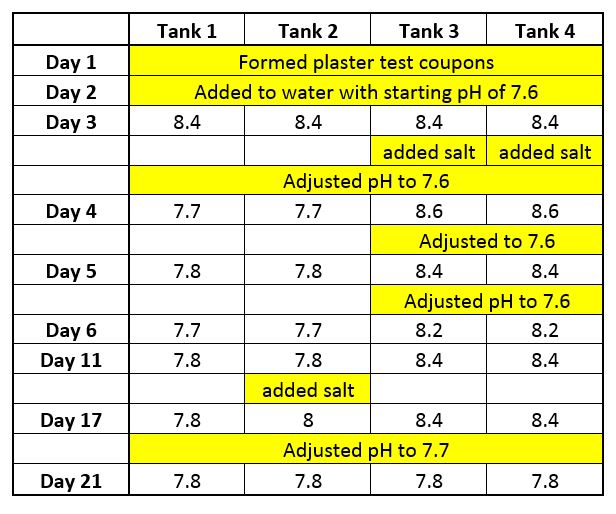
On Day 1, four good quality plaster coupons were formed and allowed to harden.
On Day 2 the coupons were placed into four separate water tanks. The water in all four tanks were balanced to the same parameters; Temp = 70°F, pH = 7.6, TA = 160 ppm, CH = 200 ppm. The tanks were capped to slow down carbon dioxide out-gassing. (When water loses CO2, the pH rises with no change in TA).
Day 3, after 24 hours in water, the pH of the water was recorded and in all four tanks the pH rose to 8.4, indicating that some hydroxide from the plaster surface had been dissolved away and into solution. (Note that this occurred even in balanced water). The pH would have risen even higher if the beginning TA of the water was lower to around 80 ppm.
Then Tanks 3 and 4 had 3000 ppm of salt added. Acid was then added to all water tanks to lower the pH to 7.6.
On Day 4, the pH in Tanks 1 and 2 rose slightly to 7.7. But in Tanks 3 and 4, the pH had risen much higher to 8.6, indicating a significant effect on the plaster surface material. Acid was added to Tanks 3 and 4 to lower the pH back to 7.6.
On Day 5, the pH in Tanks 1 and 2 was 7.8, but in Tanks 3 and 4 the pH rose to 8.4. Again, the pH raised more in the Tanks that had the salt added. Acid was added again to all Tanks and lowered to pH 7.6.
On Day 6, the pH in Tanks 1 and 2 was 7.7, but in Tanks 3 and 4 the pH was 8.2. The pH was not adjusted downward.
On Day 11, the pH in Tanks 1 and 2 was 7.8; the pH in Tanks 3 and 4 was 8.4. At this time, 3000 ppm of salt was added to Tank 2.
On Day 17, the pH of Tank 1 was 7.8; the pH of Tank 2 was 8.0, the pH of Tank 3 and 4 was still at 8.4. This data shows that the salt added to Tank 2 after 11 days had a slight effect on the plaster coupon in comparison to Tank 1. The pH in all tanks were lowered to 7.7.
On Day 21, the pH of all tanks was identical at 7.8.
(pH was determined using a Kruger & Eckels Model 100 pH meter coupled to a wide range combination electrode with a silver/silver chloride reference. Samples are tested at room temperature (25ºF) unless otherwise noted. Accuracy +/- .05 pH units)
