by Que at onBalance
Remember sitting in high school math class, griping along with the other students about why we needed to learn all that stuff? Saying “When will I ever use this in real life?” Well, here we are, pool owners, and we find that to dose a swimming pool properly we need to use that stuff after all!
To determine the volume of a square, rectangular, oval or kidney pool a service tech needs to apply one of several algebraic formulas. Fortunately, they are not too complicated:

Rectangular or square: length x width x average depth x 8.3 = volume in gallons

Round: 3.14 x radius x radius x average depth x 7.5 = volume in gallons
Oval: 3.14 x 1/2 length x 1/2 width x average depth x 7.5 = volume in gallons
The funny part comes when you need to find the volume of a freeform pool:

The instructions essentially say “divide the pool into rough geometric shapes, solve for those shapes, add the numbers together, and you will be somewhere in the ballpark.” Not only can this be difficult on some pools, but determining the average depth for these kinds of pools is an added challenge. Obviously a better way needed to be found.
Much to our surprise, as we at onBalance were working on correcting industry alkalinity dosage charts, one of my old math lessons jumped back at me: if A x B = C, then C ÷ A = B! Or in other words, since a specific amount of acid or base added to a specific volume of water results in a given alkalinity shift, then we can rearrange those variables and say that the amount of a chemical added, divided by the change in alkalinity, equals the water volume!
The one tricky part about doing this at poolside is that to get an accurate answer you need to be able to test alkalinity to within one part per million, rather than the common 10 ppm precision of most poolside, and even retail store test methods. So get a glass, and add precisely 10 times as much pool water as is normally used for an alkalinity test sample. Then add enough color indicator to see it well. (Using more or less color indicator will not change the results of the test.) Then start adding the drops of titrant – but now each drop is 1 ppm instead of 10. This will obviously use up more titrant, but you only do this once per pool, and we promise you the test kit supplier won’t mind if you buy refills of their solutions!
Here is the simple field method we use:
- Take a water sample of 10 times the normal amount from the pool for testing.
- Add acid, soda ash, or another chemical which alters total alkalinity to the pool. Add enough to make a noticeable shift in total alkalinity – at least 10 to 20 ppm.
- Write down exactly how much you used.
- Wait for the chemical to completely blend throughout the water. (This time will vary from 45 minutes to several hours depending on the method of addition, the circulation, etc.)
- Take a second 10X water sample from the pool.
- Test the total alkalinity of the two samples.
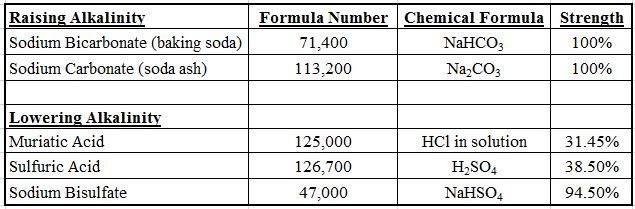
- Find the formula number for the chemical you used from the list below.
- Apply the following formula (using the appropriate formula number depending on which chemical was used):
(formula number)(amount of chemical used in quarts or pounds)
—————————————————————————— = Pool Volume
(Number in ppm that the TA changed)
Here are a couple of examples. First, if you add 5 pounds of sodium bicarbonate (NaHCO3, or baking soda) to a pool, and the starting TA was 100 ppm and the ending TA was 116 ppm, then:
(71,400 for baking soda)(4 pounds)
—————————————— = a 19,000 gallon pool
(115 – 100, or 15 ppm)
Or if you add two quarts of acid to a pool and the alkalinity drops 12 ppm:
(125,000 for muriatic acid)(2 quarts)
——————————————— = a 24,000 gallon pool
(120 – 110, or 10 ppm)
Actually, the volumes in the first example came out to be 19,040, but of course that is too precise – round off to the nearest 1000 gallons. The margin of error should be around 5 %